Hexafluoro-2-butyne
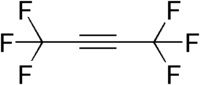 | |
 | |
| Names | |
|---|---|
| IUPAC name
1,1,1,4,4,4-Hexafluoro-2-butyne | |
| Other names
HFB | |
| Identifiers | |
| 692-50-2 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 62855 |
| ECHA InfoCard | 100.010.667 |
| PubChem | 69654 |
| RTECS number | ES0702500 |
| |
| |
| Properties | |
| C4F6 | |
| Molar mass | 162.03 g/mol |
| Appearance | Colorless gas |
| Density | 1.602 g/cm3 |
| Melting point | −117 °C (−179 °F; 156 K) |
| Boiling point | −25 °C (−13 °F; 248 K) |
| Insoluble | |
| Structure | |
| 0 D | |
| Hazards | |
| Main hazards | Toxic gas |
| R-phrases | R12 R23 |
| S-phrases | S16 S33 S45 |
| Related compounds | |
| Related compounds |
Dimethyl acetylenedicarboxylate Hexachlorobutadiene Acetylene |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Hexafluoro-2-butyne is the fluorocarbon with the formula CF3C≡CCF3. HFB, as it is known also, is a particularly electrophilic acetylene, hence a potent dienophile for Diels–Alder reactions.[1][2]
It is prepared by the action of sulfur tetrafluoride on acetylenedicarboxylic acid and the reaction of KF with hexachlorobutadiene.
References
- ↑ Essers, M.; Haufe, G. "Hexafluoro-2-butyne” in Encyclopedia of Reagents for Organic Synthesis (Ed: L. Paquette) 2004, J. Wiley & Sons, New York. doi:10.1002/047084289.
- ↑ E S Turbanova, A A Petrov (1991). "Perfluoroalkyl(aryl)acetylenes". Russian Chemical Reviews. 60 (5): 501–523. doi:10.1070/RC1991v060n05ABEH001092.
This article is issued from Wikipedia - version of the 4/30/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.